 
T 1 to t n are the time intervals at each of the sample points. T 1 to T N are the temperature at each of the sample points in kelvin. R is the gas constant (in J mol -1 K -1). ΔH is the activation energy (in kJ mol -1). T K is the mean kinetic temperature in kelvin. When establishing the mean kinetic temperature for a defined period, the formula of J. Therefore you can easily convert between the two temperature scale by a simple. The mean kinetic temperature is higher than the arithmetic mean temperature and takes into account the Arrhenius equation. Part 2 More advanced topics on the ideal gas laws, calculations, kinetic. Mean kinetic temperature: a single derived temperature that, if maintained over a defined period of time, affords the same thermal challenge to a drug substance or drug product as would be experienced over a range of both higher and lower temperatures for an equivalent defined period. 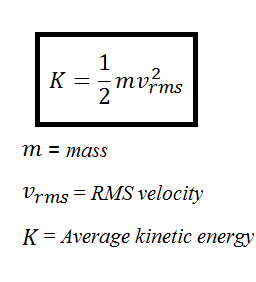 
The mean kinetic temperature is the total influence of temperature on an object or product over a certain period of time.Įxtract from the ICH Topic Q 1 A (R2): Stabilty Testing of new Drug Substances and Products: Calculation impact of arithmetic mean kinetic value which equation, the quantity of the excel function not the temperature. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
March 2023
Categories |
 RSS Feed
RSS Feed
